
2022 Entry Clinical Fellowship
Development of peripheral nerve specific biomarker assays: from in vitro neuropathy models to clinical validation
Guillain-Barré syndrome (GBS) and chronic inflammatory demyelinating polyradiculoneuropathy (CIDP) are conditions where the immune system, the body’s natural defence against illness and infections, mistakenly attacks and damages the nerves, causing progressive paralysis. In severe cases, people can lose their ability to walk, swallow and breathe. About 1 in 20 people with GBS die, and many are left with long-term disability.
Treatments for these diseases are only partially effective. Part of the problem is that we do not have biomarkers (naturally occurring molecules) to diagnose and assess response to treatment, as the amounts released by the damaged nerves are tiny and extremely difficult to measure.
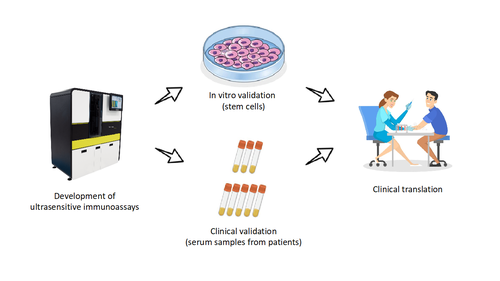
Recently, very powerful machines called “single molecule analysers” (SiMoA) have enabled us to measure biomarkers at 100-2000 times lower concentrations than before (equivalent to being able to detect a teaspoon of sugar dissolved in a full-sized Olympic swimming pool).
In my project, I will study two potential biomarkers of nerve damage, peripherin and periaxin, and develop SiMoA assays to measure them in the blood. I will grow human peripheral nerve cells in a dish in the laboratory using stem cells, which have a special ability to turn into different cell types, including those of the nerves. I will then artificially damage these cells and measure peripherin and periaxin released after damage. Finally, I will measure the biomarkers in the blood of people with inflammatory neuropathies.
This work will positively impact on both clinical and research aspects of the inflammatory neuropathies. Clinically, peripheral nerve specific biomarkers will assist with diagnosis, prognostication, monitoring of disease activity and titration of treatment. On a research level, peripherin and periaxin could be used as surrogate intermediate outcome measures, improving the efficiency of clinical trials and accelerating advances in care.
Publications
Ultrasensitive assay technology and fluid biomarkers for the evaluation of peripheral nerve disease
Journal of Neurology, Neurosurgery & Psychiatry. 2023 Oct 11:jnnp-2023-332031. doi: 10.1136/jnnp-2023-332031
2023
AL amyloidosis presenting with isolated lumbosacral radiculoplexus neuropathy
Practical Neurology 2023 Jul 17:pn-2023-003788. doi: 10.1136/pn-2023-003788
2023
Reply: Peripherin is a biomarker of axonal damage in Guillain Barré syndrome: a pathophysiological annotation
Brain 2023 Aug 30:awad276. doi: 10.1093/brain/awad276
2023
Peripherin is a biomarker of axonal damage in peripheral nervous system disease
Brain. 2023 Jul 12:awad234. doi: 10.1093/brain/awad234
2023
Treatment of CIDP
Practical Neurology. 2023 Feb 1;23(1):46-53, http://dx.doi.org/10.1136/pn-2021-002991
2023